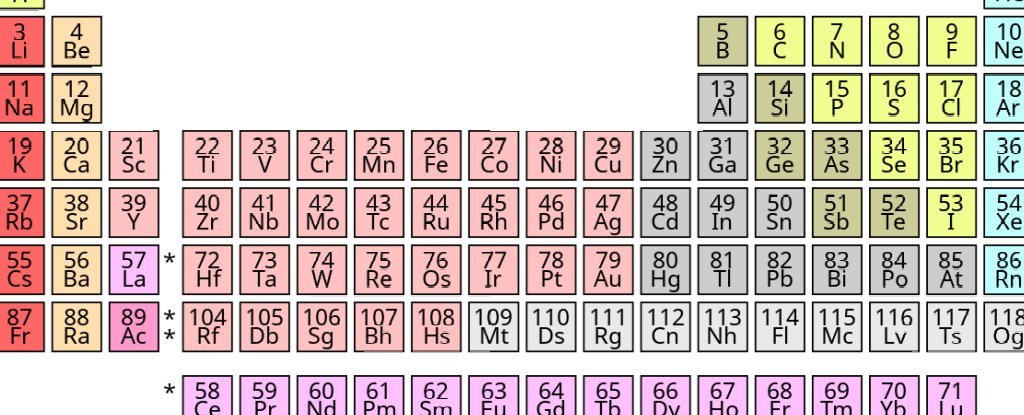
Periodic table of elements, mainly created by a Russian chemist, Dmitry Mendeleev (1834-1907), its celebrated 150th Anniversary Last year. Its importance as an organizing principle in chemistry is hard to overestimate – all emerging chemists are familiar with it from the very beginning of their education.
Given the importance of the table, one might apologize for assuming that the order of the elements is no longer subject to discussion. However, two scientists in Moscow, Russia, recently published a Suggestion for a new order.
Let us first look at how the periodic table was developed. By the end of the eighteenth century, chemists were clear about the difference between an element and a compound: the elements were chemically indistinguishable (e.g. hydrogen and oxygen), but the compounds contained two or more elements that were completely different from their constituent elements.
At the beginning of the nineteenth century, it was there Good circumstantial evidence For the existence of atoms. By the 1860s, known elements could be listed in terms of their relative atomic mass – for example, hydrogen 1 and oxygen 16.
Simple lists are, of course, monotonous. Chemists knew that some elements had similar chemical properties: for example lithium, sodium, potassium or chlorine, bromine and iodine.
Something seems to be repeating, and by placing chemically similar elements on top of each other, a two-dimensional table can be constructed. The periodic table was born.
Importantly, Mendeleev’s periodic list was empirically based on the observed chemical similarities of certain elements. Until the beginning of the twentieth century, after the structure of the atom was established, with the development of quantum theory, a theoretical understanding of its structure would emerge.
The elements are arranged by atomic number (the number of positively charged particles called protons in the atomic nucleus) rather than the atomic mass, but are still chemically similar.
The latter, however, follow the arrangement of electrons called “shells” at regular intervals. By the 1940s, most textbooks included a periodical, as we see today, as shown in the picture below.

It is understandable that this would be the end of the matter. However, this is not the case. A simple search of the internet will reveal All kinds of versions Of the periodic table.
There are short versions, longer versions, circular versions, spiral versions, and 3D versions. Many of these are different ways of transmitting the same information, but disagreements remain over where certain elements should be placed.
The exact location of certain elements depends on the specific features we want to highlight. Therefore, a periodic table that emphasizes the electronic structure of atoms will be different from tables with important chemical or physical properties.
These versions are not very different, but there are some elements – for example hydrogen – that can be placed completely differently depending on the particular property you want to highlight. Some tables place hydrogen in group 1, while others sit above group 17; It is even on some tables In a group of your own.
However, more radically, the arrangement of the elements can be considered in a very different way, which does not contain atomic numbers or reflect the electronic structure – reversing to a dimensional list.
New proposal
The latest attempt to arrange the elements in this way Recently published Journal of Physical Chemistry Scientists Saheed Alhari And Artem Oganov.

Their approach, Based on the previous work of others, Is to assign each component a Mendeleev number (MN).
There are several ways to obtain such numbers, but the latest study uses a combination of two basic quantities that can be measured directly: the atomic distance of an element and a property. Electro Negativity This explains why an atom attracts electrons to itself.
If one orders their components their MN, let the nearby neighbors be surprised, they have a similar MN. But for further use it goes a step further and builds a two-dimensional grid based on the MN of the so-called “binary compounds”.
These are compounds consisting of two components, sodium chloride and NaCl.
What is the benefit of this approach? Importantly, it can help predict the characteristics of binary compounds that have not yet been manufactured. It is useful in the search for new materials needed for future and current technologies. Over time, this will expand into compounds with two elemental components.
Given the periodic table shown in the figure below, a good example of the importance of searching for new materials can be appreciated.

This list highlights not only the relative abundance of elements (each component has a larger box, there is more of it), but also the distribution issues relevant to technologies that have become ubiquitous and indispensable in our daily lives.
Take mobile phones for example. All the components used in their manufacture have been identified using the phone icon, and you can see that many of the required components are scarce – their future distribution is uncertain.
If we are developing replacement materials that avoid the use of certain components, the insights gained from ordering the components may be valuable in that search.
150 years later, periodicals are not only an important educational tool, but also continue to be useful to researchers in the search for essential new materials. We should not think of new versions to replace the previous illustration. Having different lists and lists helps to increase our understanding of how components work.
Nick Norman, Professor of Chemistry, University of Bristol.
This article has been republished Conversation Under the Creative Commons license. Read Original article.

Prone to fits of apathy. Unable to type with boxing gloves on. Internet advocate. Avid travel enthusiast. Entrepreneur. Music expert.